Growing up, Jada White struggled to keep a consistent friend group at school because she spent so much time away from the classroom.
Severe pain caused by sickle cell disease continually sent her to the hospital for blood transfusions and treatment for pain or infections.
“It was hard to make friends and socialize because I would miss half of the whole school year,” White said.
The people she met at Spectrum Health Helen DeVos Children’s Hospital became her social circle instead.
“I knew everybody at the hospital. It’s basically my second home,” said White, 24, of Grand Rapids, Michigan.
When she was young, White coped by making a game of her monthly blood transfusions, telling herself, “Hey, the poke is going to hurt, but I get a sucker or a candy out of the toy box.”
Hospital admissions became more frequent during White’s high school years. Sarah Smith, a teacher with the Dick and Linda Antonini Hospital School Program, served as a liaison with her classroom teachers and made sure she got her homework done.
“It was really hard, but because of her I was able to graduate with a 4.1 GPA” in 2016, White said. “I’m glad she pushed me.”
Keeping faith
After high school, White completed an associate’s degree at Davenport University. She also worked part-time in a hospital cafeteria, but as her disease progressed, working became more difficult.
“As you get older, the pain gets worse,” White said. “It kind of, like, breaks you down.”
Sickle cell disease is an inherited blood disorder characterized by a flaw in the hemoglobin—the protein in red blood cells that carries oxygen from the lungs to the rest of the body.
In people who have the disease, the red blood cells change in form from flexible discs to stiff crescents. These sickle-shaped cells have trouble moving through the blood vessels.
As a result, the tissues don’t get enough oxygen.
The shortage of healthy red blood cells causes anemia, while the lack of oxygen causes episodes of severe pain, called pain crises.
“It’s different for everybody, but with me, my pain crisis was always in my left leg,” White said, describing it as a constant stabbing pain.
In the United States, sickle cell disease occurs most commonly in African Americans. About one in every 365 Black children is born with the disease. It also affects some people with Hispanic, southern European, Middle Eastern or Asian Indian backgrounds.
White long knew her chances for a cure were slim.
For years, a stem cell transplant was the only available cure for patients with severe sickle cell disease, but this required a matching donor—a sibling whose human leukocyte antigen, or HLA, tissue type perfectly matched the patient’s.
White didn’t have one.
Her only full sibling was not a match—nor did she have good odds for finding an unrelated donor through the donor registry.
Knowing that adults with sickle cell disease have a shortened lifespan and a risk for stroke, White relied on her strong faith in God and her upbeat personality to keep going.
“You have to manage it—you have to find ways to cope with it and continue being happy,” she said.
When emotions ran high, White wrote poetry, jotting down lines in the notes app of her iPhone. It became an important source of encouragement.
Clinical trial
A couple of years after high school, new hope dawned for White.
Hope came not in the form of a new treatment but in a variation on an existing treatment.
Researchers had introduced a clinical study to explore the safety and effectiveness of bone marrow transplantation using a related donor with a half-matched, or haploidentical, rather than identical HLA tissue type.
Haploidentical donors can be parents, children, siblings and sometimes cousins of the patient.
To qualify for the study, patients have to meet specific health criteria and agree to participate in two years of post-transplant tracking.
As the leader of the Spectrum Health Pediatric Blood and Bone Marrow Transplant program, Ulrich Duffner, MD, already knew White and understood her situation.
He knew that she’d been interested in stem cell transplantation but didn’t qualify under the conventional protocol.
Wanting to offer better treatment options for White and others like her, he sought approval for Helen DeVos Children’s Hospital to participate in the study.
Once authorization came through, making Spectrum Health one of 32 participating centers nationwide, Dr. Duffner discussed the clinical study in detail with White and her mother, Tammer Saffore.
“Haploidentical stem cell transplant, like other types of bone marrow transplant, replaces the red blood cells which have the sickle cell defect with cells now made by the donor’s bone marrow,” he explained.
Doctors use haploidentical transplant for patients with other diseases, but it is still unclear how well it might work in treating sickle cell disease.
“That’s part of what we have to discuss with our patients,” he said.
“Do you take more of that risk now with the hope that your further life as an adult should be much better, or do you say, ‘No, I’m worried about the risks of a transplant, and I’ll do the best treatments we can do without a transplant’?”
Saffore underwent testing and learned she would be a good haploidentical donor for her daughter. But because of the risks to White, Saffore had serious hesitations.
White wanted to enroll in the study, so she kept encouraging her mom to agree.
Eventually she relented.
“She saw the pain in my eyes and just said yes,” White said. “It was getting too severe.”
The study’s parameters allowed White to undergo the transplant at Helen DeVos Children’s Hospital, though she had already turned 21.
So in November 2019, mother and daughter prepared for the transplant.
For White, that meant receiving reduced-intensity chemotherapy and radiation to condition her body to receive the transplanted bone marrow.
Pre-transplant conditioning prepares the body in two ways, Dr. Duffner said: It makes space in the body for the donor’s blood stem cells, and it suppresses the patient’s immune system to prevent it from rejecting the donor cells.
Part 2 of the study’s conditioning regimen comes three days after the transplant, when the patient receives an additional form of chemo called cyclophosphamide.
On White’s transplant day, her mother had her bone marrow collected under anesthesia.
Dr. Duffner then administered these donor cells to White through a central line, like a blood transfusion, so they could find their way to their new home.
“The blood stem cells, they are smart—they know where to go with the bloodstream in our body, to find the niche in the bone marrow,” Dr. Duffner said.
Typically after about two weeks, he said, “we see the first cells now coming from the donor bone marrow.”
When the clinical study concludes in late 2024, doctors will learn whether the overall results will lead to a new standard of care for a significant group of patients with severe sickle cell disease.
If the conclusion is positive, “that could really become a life changer and a game changer down the road,” said Laura Paulsen, RN, White’s clinical research nurse.
Post-transplant recovery
The transplant itself went smoothly for White.
She faced some other challenges in her recovery, which required visits to her care team.
“She was in the hospital, back and forth, for some months,” according to Sarah Straveler, RN, White’s transplant coordinator. “And when she was not here, she was in our (outpatient) clinic three or four times a week.”
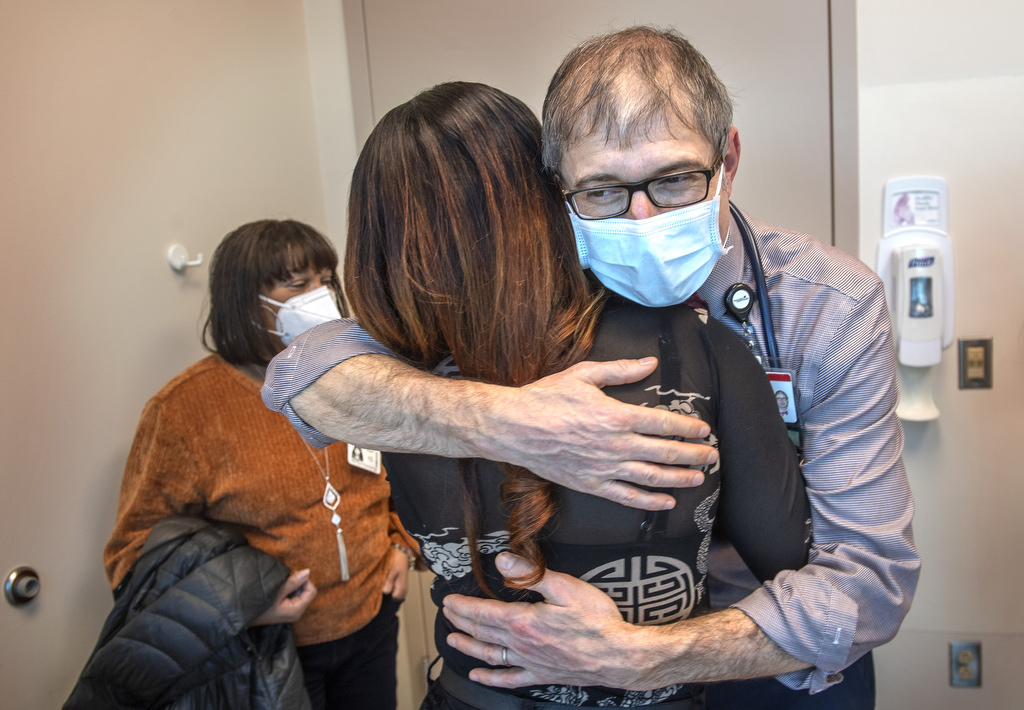
Yet, two years later, Dr. Duffner is pleased with White’s positive outcome. Her bone marrow is stable, she no longer takes immune suppression medication and she’s considered cured of sickle cell disease.
The issues that remain for her—including some nerve and bone damage—are mainly after-effects of the disease in her body, not of the transplant, he said.
Dr. Duffner has reason to hope these things can continue to improve.
“As more and more time goes by with the blood cells not having the disease anymore, we have seen with other patients that more and more recovery from sickle cell disease can happen,” he said.
“These systems, they can recover.”
That’s great news for White, who after spending months in outpatient rehab to regain strength and mobility, has returned to her cafeteria job.
She has only just begun to imagine what her future might hold.
“Now that I’m cured, I don’t know what to do with my life,” she said, laughing.
“I want to be humble, but at the same time, I want to shout, you know? To tell everybody there’s still hope in the world.”
When she takes time to dream, she envisions working with children.
“I always wanted to be a nurse anesthetist or somebody that worked with pediatric patients, because of how I was brought up at Helen DeVos (Children’s Hospital),” she said.
“My hospitality goes towards children … which means my obligation to them is to let them know that somebody’s there, or somebody went through what they’re about to go through.”
White is not only the first person at Helen DeVos Children’s Hospital to enroll in this clinical study. She is the children’s hospital’s first patient with sickle cell disease to receive donor blood stem cells.
Looking back on her participation in the study, White feels deep gratitude for the care provided by her health team.
She’s also grateful for her mother’s gift and her grandmother’s daily visits.
“The only thing that got me through this experience was the love of my family and God,” she said. “With the help of the doctors, I just feel like my life is a blessing.”
















 /a>
/a>
 /a>
/a>
 /a>
/a>
So happy to read this! Proud of you, Jada!
Congratulations to you Jada. You are part of history and the future. This is a mark that cannot be erased. Praise God for using the sciences for your healing!!
So proud of you Jada! Congratulations and go get your dreams! You are amazing and you can!!
So thankful for doctors and nurses! Jade, God will open a door for you. Just listen.
I have a 2 nephews who has suffered with sickle cell anemia all all their lives. The 31 year old has had it the worst and is constantly in and out of SH hospital due to pain from the crisis and requiring blood transfusions. My heart breaks for him and I feel so bad because his mother, my sister passed away in 2018 at the age of 58 from small cell lung cancer. She was always with him when he was hospitalized and always hoped that someday there will be a cure for this disease. Glad for Jada may God continue to bless her!
This is a beautiful story. Sounds like she has overcome obstacles most can not even imagine. Happy to hear to the amazing progress.