If it sounds too good to be true, it probably is.
A new study warns consumers to steer clear of clinics marketing treatments and therapies that claim to heal through use of stem cells.
Researchers from the University of California, Davis, and the University of Minnesota conducted a detailed examination of U.S. businesses promoting stem cell therapies, and they found a whopping 570 of these clinics and spas across the country—including eight in Michigan.
The clinics market these treatments for cosmetic purposes and an array of disorders or diseases, including Alzheimer’s, autism, cerebral palsy, muscular dystrophy and Parkinson’s—all without a scientific consensus on their safety or effectiveness, and without the blessing of the U.S. Food and Drug Administration.
“This commercial activity generates a host of serious ethical, scientific, legal, regulatory and policy concerns,” according to the study, published in the journal Cell Stem Cell.
Snake oil
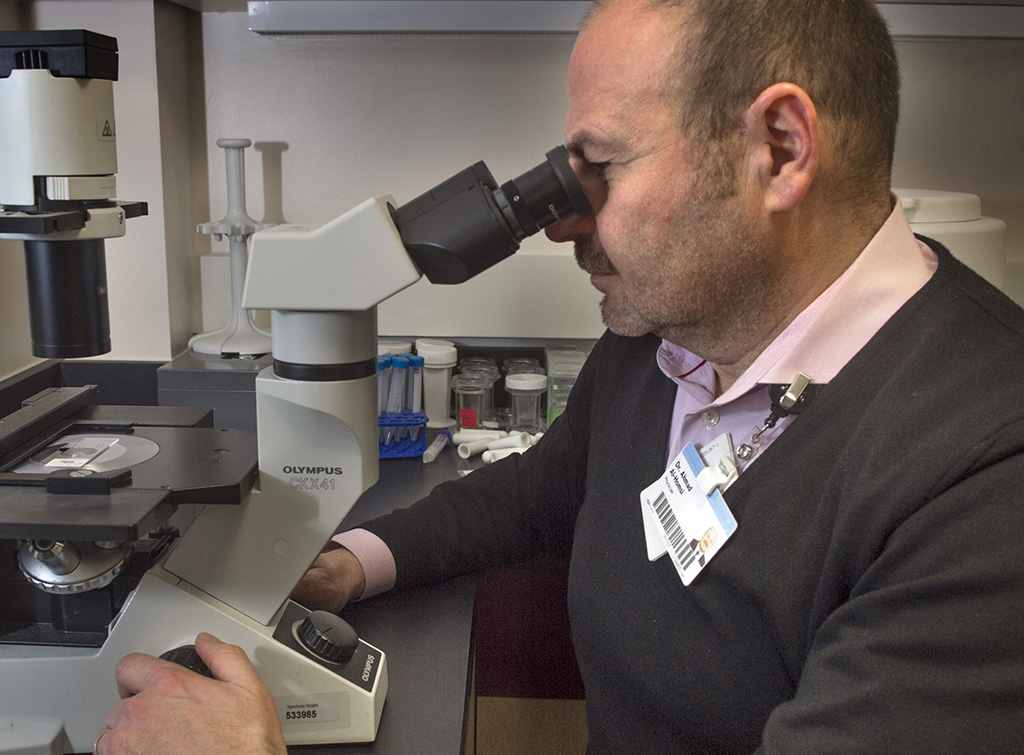
Samer Al-Homsi, MD, a blood and marrow transplant specialist with Spectrum Health Medical Group, shares the concerns of the study’s authors.
His main fear is people could be led astray by clinics using the term “stem cell.”
The stem cell transplantation that Dr. Al-Homsi and his colleagues at the Spectrum Health Cancer Center use to treat certain cancers has nothing to do with what these direct-to-consumer businesses purport to offer, he said.
“What we do is vigorously regulated by many agencies, including the FDA,” Dr. Al-Homsi said. “It’s scientifically tested and scientifically sound and undergoes the most rigorous scientific processes to prove its efficacy.
“So we should really not mix … these ‘snake oils’ and what we do here,” he said.
Dr. Al-Homsi’s concerns about unregulated treatments are mainly ethical: How can clinicians in good conscience risk using products and techniques that aren’t regulated or scientifically proven?
“Certainly most of them have not been subjected to any sort of scientific examination by any acceptable scientific criteria … to ensure safety,” the doctor said. “Every time you talk about potentially living cells in there, that’s a concern … that you can transmit infectious agents.”
He added: “I worry about some of these products being from animal origin and not clearly defined.”
People should be very cautious, particularly if they’re considering using these treatments for serious issues such as Alzheimer’s or autism, he said.
FDA input
Confusion sometimes arises from lack of understanding.
People have heard about researchers using stem cells to regenerate organs, for instance, but that’s just it—it remains in the realm of research.
The FDA has approved only one stem cell product. It’s for patients who have problems with their body’s blood-forming system.
The agency is updating its guidelines on the regulation of human cells, and it has scheduled a public hearing on the topic in September.
In a consumer update, the FDA expressed concern “that the hope that patients have for cures not yet available may leave them vulnerable to unscrupulous providers of stem cell treatments that are illegal and potentially harmful.”
What these providers are doing, in Dr. Al-Homsi’s judgment, “is basically selling hope.”
As for cosmetic or anti-aging stem cell treatments, the message is the same: Buyer beware.
Of course, if someone insists on getting a so-called stem cell facelift, so be it. It’s just a waste of money, Dr. Al-Homsi said.









 /a>
/a>
 /a>
/a>
 /a>
/a>