Once upon a time, there was a pulmonologist named Dr. John Schuen.
He cared for kids with cystic fibrosis, an inherited disease that causes thick mucus to gather in the lungs and digestive tract.
Often, he was a very sad pulmonologist.
You see, this was the early 1990s. Dr. Schuen had just finished his medical training and while he was excited to make a difference in the lives of kids with cystic fibrosis, he knew that the research pointing to new treatment frontiers was still just theoretical.
In reality, all he and other doctors could do was to treat the symptoms of the disease, not the cause. Unfortunately, many of his patients never reached adulthood.
“Life expectancy back then was just horrible,” Dr. Schuen said. “Teens didn’t even plan for college or having a career.”
Today, he’s a much happier pulmonologist. Hope is no longer theoretical. It’s a reality.
“Now I’m invited to graduation ceremonies, not funerals,” he said. “And I tell parents to expect that their infant with CF can have a bright future. They can and should plan for college, career and family—the things we take for granted.”
Giddy-worthy news
The week of July 6, 2015, Dr. Schuen was downright giddy with joy and excitement.
“It’s a banner week for people with CF,” he said.
Why?
The U.S. Food and Drug Administration approved Orkambi (lumacaftor and ivacaftor) to treat cystic fibrosis in patients who have inherited two copies of a specific gene mutation from their parents.
Nearly half of the estimated 30,000 individuals with cystic fibrosis in the U.S. have the two copies of the F508del gene mutation and could eventually benefit from this new therapy.
“This is a real game-changer for our patients,” Dr. Schuen said. “Orkambi is the first precision drug that improves key symptoms and targets the underlying cause of CF.”
Already, he and his team at the Spectrum Health Helen DeVos Children’s Hospital Cystic Fibrosis Clinic have identified patients who may be eligible for the new drug.
And while the FDA approval is for patients ages 12 and older, Dr. Schuen noted that other studies are already planned for younger children.
What’s so special about Orkambi?
It combines two proven drugs for a more effective two-step treatment approach. First, the lumacaftor helps move a defective protein to its proper place at the cell surface. Then, ivacaftor increases the activity of the protein once it’s there to help thin the thick mucus that builds up in the lungs of people with cystic fibrosis.
The result: improved lung function and significantly reduced rates of inflammation and infection.
“Patients can breathe easier, expect to spend less time in the hospital and keep their lungs healthier, longer,” Dr. Schuen said. “It’s just awesome news!”
Life-changing options
Indeed it is for patients like Katriona VanDoorne, a 2015 graduate of Grand Haven High School. According to her mom, Kelly, Katriona’s on track to receive Orkambi soon.
“I hope this is life-changing for her and for her future,” Kelly said. “I want for her not to be so exhausted, to have some normalcy as she becomes an adult. I now have a sense of even bigger, better things coming for her.”
What does Katriona plan next?
“To hang out, babysit and work at Michigan Rag, a store in downtown Grand Haven,” Katriona said, in typical teenage fashion, of her immediate summer plans.
She also talked about photography, a hobby of hers. She got a new camera as a graduation gift. And she may take some classes at a community college this fall. Her mom mentioned she might want to run a small store someday.
What’s clear is that all options are open for her. Just like any other teenager.
With the median life expectancy today for CF patients at 42 years, and new treatments like Orkambi transforming CF care, “all bets are off” on how long they’ll live, Dr. Schuen said.
“It’s just so cool where we are today, with all the options available to treat CF that just weren’t around 20 years ago. Medicines like Orkambi just add to the excitement,” he said. “Who knows where patients like Katriona may be going in the next 20 years.”
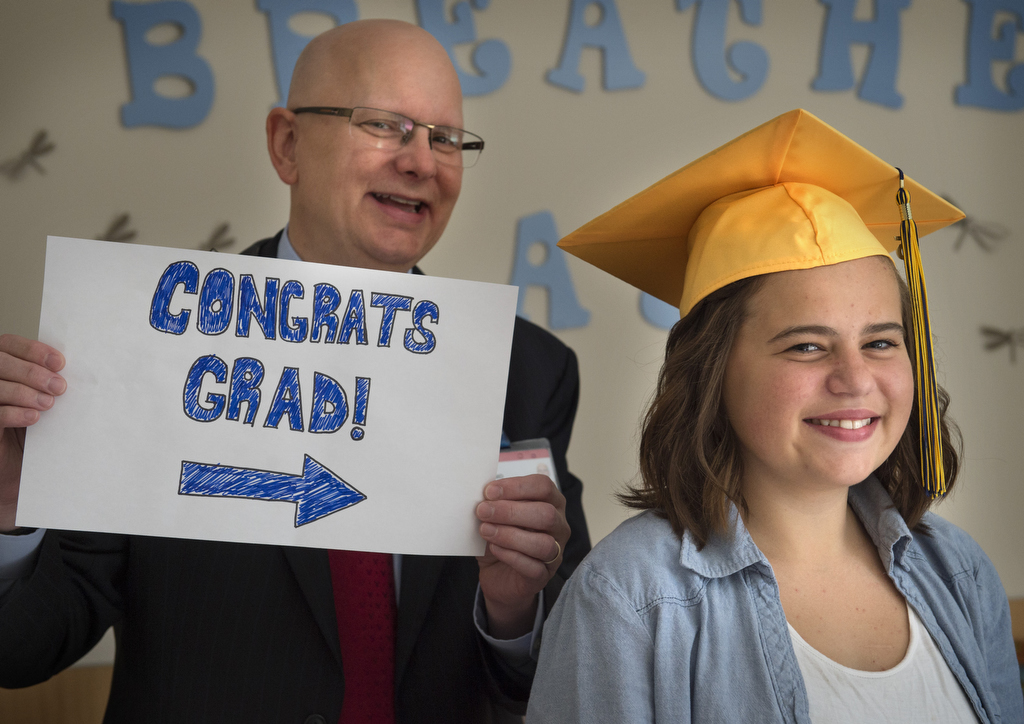










 /a>
/a>
 /a>
/a>
 /a>
/a>
What a relief for CF patients.