Ever since she moved back to Kalamazoo, Michigan, from Las Vegas two years ago to be closer to her children and grandchildren, Julia Sheppard, 61, has used a motorized cart when shopping for groceries.
Her diseased lungs made walking the aisles too difficult.
Shortness of breath bothered her every day, sapping her energy and limiting her activities.
But in January 2021, for the first time in recent memory, Sheppard did her grocery shopping on foot.
“I just went to the store—and usually I get in a buggy—but I went to the store and was pushing a cart through the store,” she said.
“I’m able to move around a lot better, and it hasn’t even been a month yet.”
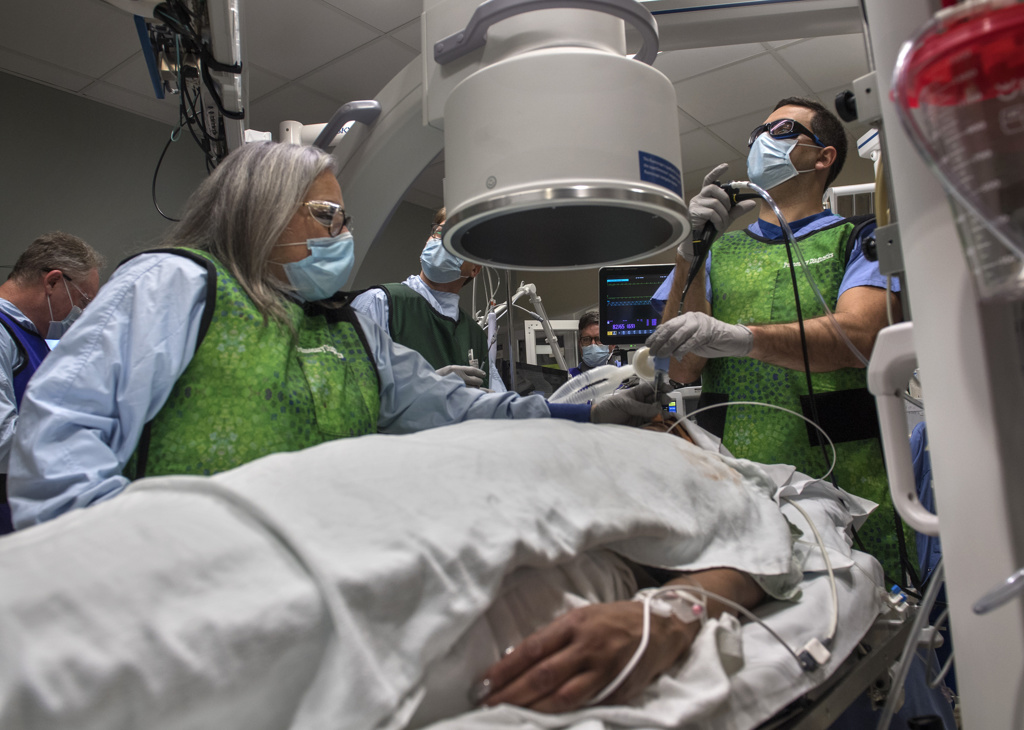
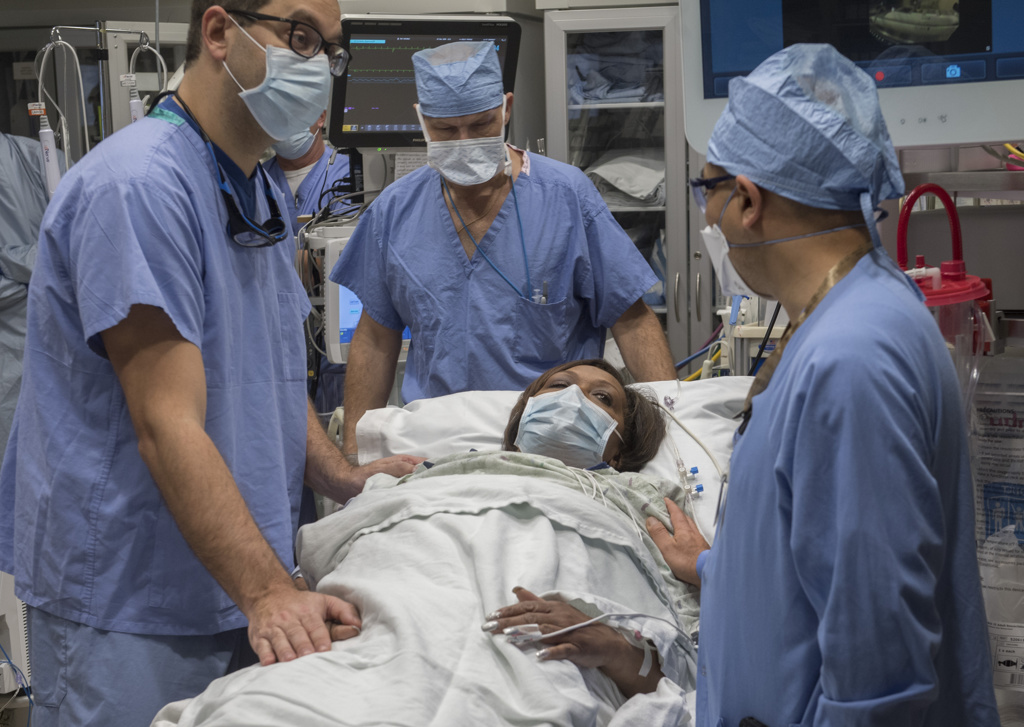
In fact, it had been only three weeks—to the day—since Sheppard underwent an innovative new pulmonary procedure at Spectrum Health Butterworth Hospital.
Three weeks since she became West Michigan’s first participant in a new clinical trial for patients with chronic obstructive pulmonary disease, or COPD, an umbrella term for diseases such as emphysema.
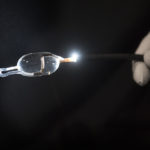
The research trial involves a one-hour outpatient procedure that uses radiofrequency ablation—targeted high-frequency energy—to damage the nerves of the lungs’ two primary airways. These hypersensitive nerves can trigger airway tightening and mucus production, making it difficult for patients to breathe.
By deactivating the airway nerves, the study aims to prevent COPD exacerbations—sudden flare-ups that can increase the risk of infection, land patients in the hospital and create additional lung damage.
Spectrum Health is one of less than two dozen clinical locations across the United States handpicked to participate in the Airflow-3 trial, sponsored by Nuvaira Inc.
Radiofrequency ablation
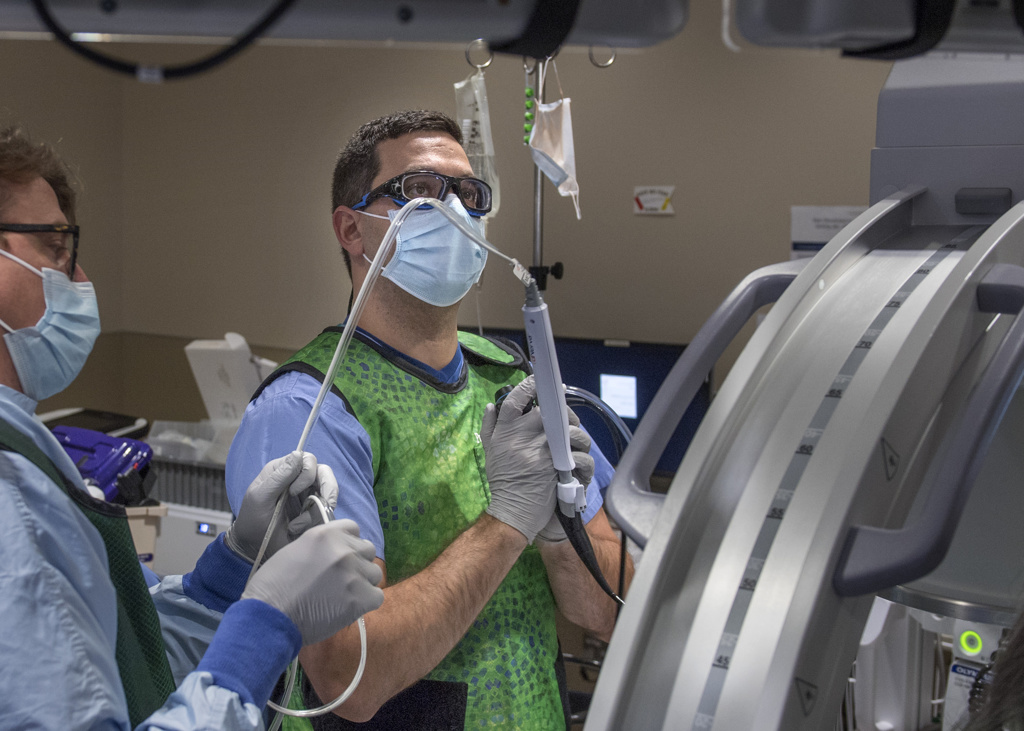
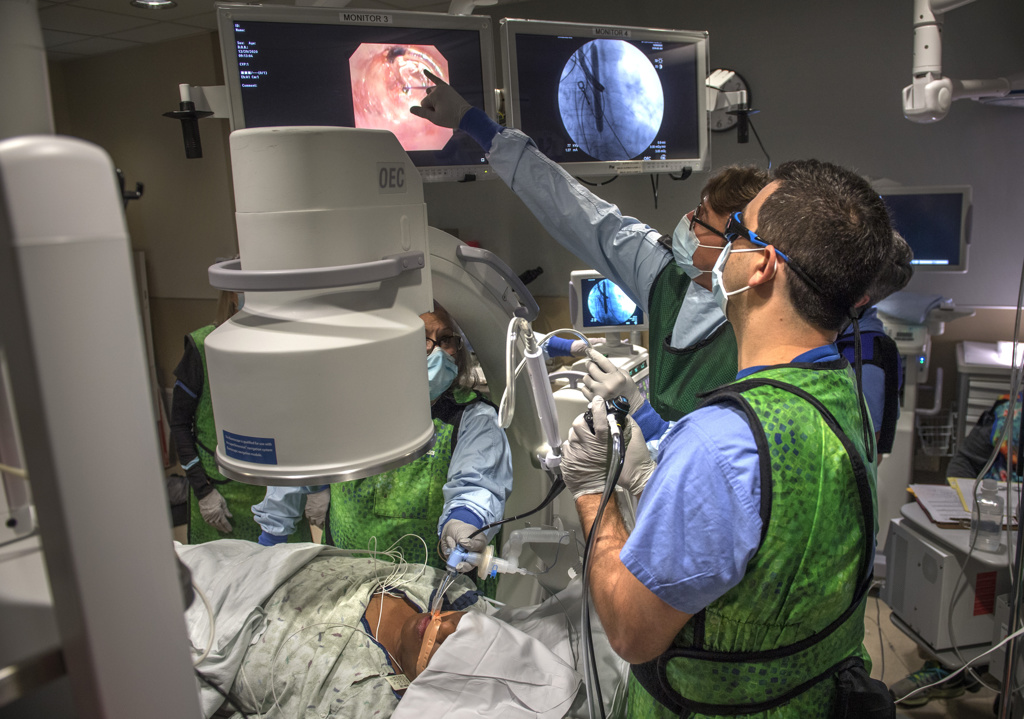
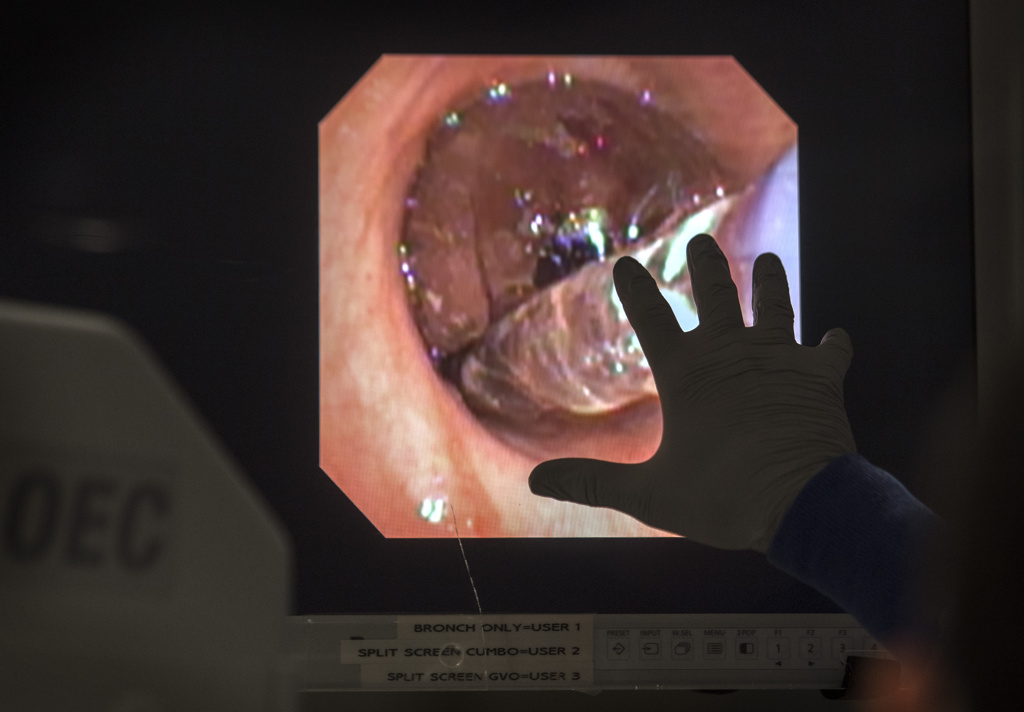
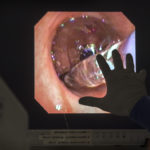
The trial procedure uses a thin, flexible scope, fed through the patient’s mouth and down to the lungs, in tandem with a specialized catheter that delivers the radiofrequency energy.
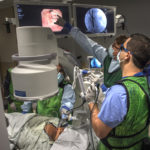
“It’s a very precise probe that gets placed in a very specific position within the airpipes, or bronchi … in order to damage the nerves that make airpipes shrink and collapse,” said Gustavo Cumbo-Nacheli, MD, a Spectrum Health interventional pulmonologist who collaborated with the system’s institutional research leaders to secure approval for the Airflow-3 trial at Butterworth Hospital.
“Over the course of time, since the nerves are not making the bronchi upset, we are expecting to see less and less flaring of their emphysema, therefore allowing patients to be symptom free.”
The earlier trial phases, Airflow-1 and Airflow-2, took place mainly in Europe and yielded promising results, Dr. Cumbo-Nacheli said.
The U.S.-based phase sets out to investigate whether this procedure, in reducing the number of COPD flare-ups, can safely deliver both short-term and long-term benefits.
“Does this procedure impact not only quality of life, but also life expectancy? That’s exactly what we are studying,” said Dr. Cumbo-Nacheli, who is the only Spectrum Health provider trained to perform the trial procedure.
He anticipates the answer will be yes because, he said, “flares are the single most important factor that impacts life expectancy” for COPD patients.
The impact of this disease is large and costly, Dr. Cumbo-Nacheli said.
COPD, which affects 16 million adults in the United States, is the third leading cause of death by disease, according to the American Lung Association.
Once the multiyear Airflow-3 study wraps up, the U.S. Food and Drug Administration can evaluate the results and decide whether to make it widely available.
Doctors have high hopes for this trial because they have so few options for treating COPD patients, according to Maximiliano Tamae Kakazu, MD, a Spectrum Health pulmonologist who partners with Dr. Cumbo-Nacheli in screening patients for admission to the Airflow-3 trial.
Dr. Tamae Kakazu follows up with trial participants for a year or more after the procedure to track their progress.
“We are limited with the interventions we can do to try to improve quality of life,” Dr. Tamae Kakazu said.
“We provide these interventions to all of our patients—inhalers, vaccination, smoking cessation counseling and, for those who qualify, also pulmonary rehabilitation and other therapies—but we hope to find another intervention that can improve how patients with COPD live.”
Positive signs
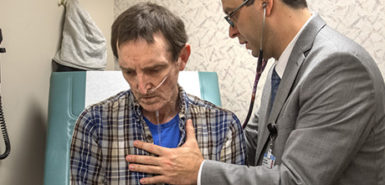
Sheppard’s doctors in Kalamazoo referred her to Spectrum Health for evaluation for this very reason—she had “maxed out on medical treatment,” Dr. Cumbo-Nacheli said.
Despite using daily medications and engaging in physical therapy, Sheppard still struggled with regular COPD flare-ups.
“I’d fight to breathe,” she said. “I’d be in the hospital between two and three times a year. A hospital stay usually is like two to three days (but) when you have a flare-up, they might last about a week or two.”
This has been her pattern for the past six years, Sheppard said. She received her COPD diagnosis eight years ago.
Recognizing Spectrum Health’s leadership in interventional pulmonology, Sheppard’s specialists hoped she would qualify for a clinical trial.
After meeting with Drs. Cumbo-Nacheli and Tamae Kakazu—and learning that she met all the trial criteria—she signed up without hesitation.
Within two weeks of her first consultation, Sheppard returned to Butterworth Hospital as West Michigan’s inaugural Airflow-3 trial patient.
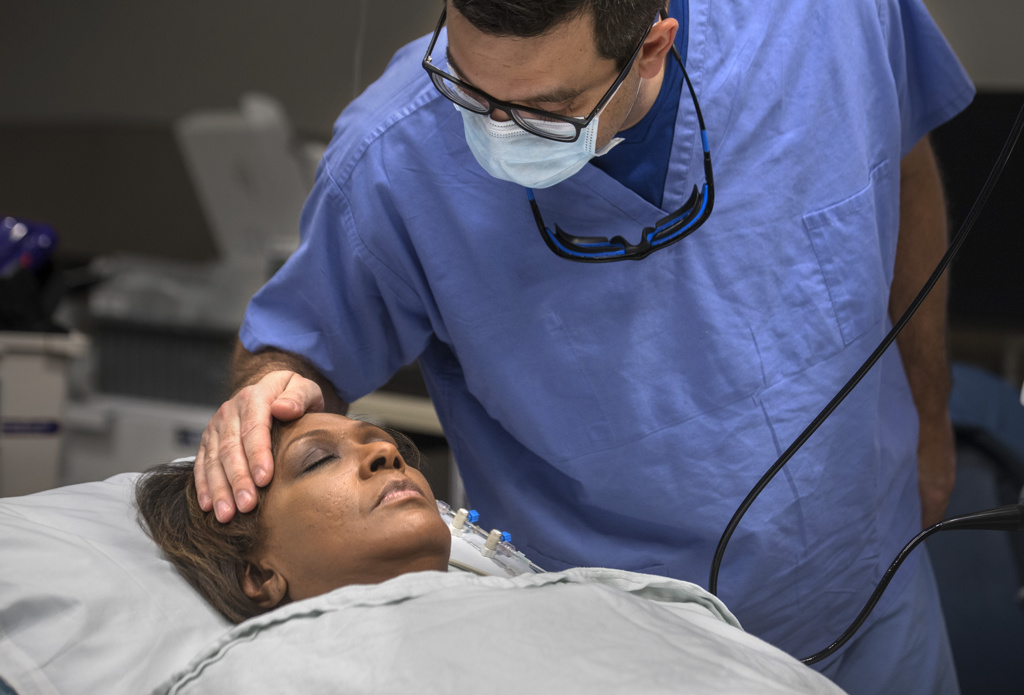
The lung denervation process, performed under sedation, proceeded “flawlessly,” Dr. Cumbo-Nacheli said.
Though the doctors cautioned that she wouldn’t likely feel positive effects of the procedure until after 30 days, Sheppard said she could tell the difference within a couple weeks.
Her breathing eased and her energy jumped.
“I’m moving around a lot better,” she said. “My breathing is a lot better. Sometimes I even forget to use my inhaler—because I’m supposed to do treatments in the morning and treatments in the evening.
“Usually, I had more bad days than good days. Now I have more good days than I have bad days. I’m feeling like a hundred percent better.”
As one of the first patients to enroll in the trial, Sheppard knew in advance she would receive the denervation treatment.
After the first two participants, however, all enrollees enter the bronchoscopy suite knowing they have a fifty-fifty chance of receiving a sham version of the procedure.
Study administrators randomly assign half of the patients to receive the ablation treatment and half to receive a sham treatment, “meaning they undergo the procedure, but no energy is delivered—so therefore there’s no cutting of the nerve,” Dr. Cumbo-Nacheli said.
Only Dr. Cumbo-Nacheli learns which patient receives which version of the procedure. By remaining in the dark, Dr. Tamae Kakazu can conduct his periodic patient follow-ups without bias or assumptions.
One year after the procedure date, patients can learn whether or not they received the ablation. Those who received the sham procedure then have the option of returning for the actual treatment.
Dr. Cumbo-Nacheli expects to enroll 15 to 20 patients in the first year. Candidate criteria include the following:
- Between 40 and 78 years old
- Take daily medication to manage COPD
- Not a current or future smoker
- Have experienced at least one COPD flare-up within the past year
‘Something good’
Bolstered by the positive early signs, Sheppard hopes to see continued health improvements.
“It seems like every day I’m feeling better and better,” she said. “And I’m hoping it gets a lot better so I can get out and do more things … like taking a walk and hanging out with my grandkids and doing stuff like that.”
Sheppard, who began smoking at age 19 and smoked a pack a day for the next 34 years, is grateful for the chance to share her story with others who may qualify for the trial.
“The way I feel now, I want to let people know that it’s something good,” Sheppard said.
“I am just so happy and so blessed that they picked me and chose me to do this.”













 /a>
/a>
 /a>
/a>
 /a>
/a>
Sounds almost to be too good to be true. Is it affordable? Am sure as a “trial” Medicare would not cover/approve payment for these services.
Thank you again, Dr. Gus
I read this and made me feel hopeful as I am having this treatment next week in the uk and was trying to find someone who had had the treatment ,gives me hope
I had it done and they paralyzed one of my vocal cords now I can barely speak. The Dr treats you like a mass production specimen to see how many he can do in one day . He has never once called me , only his staff to ask the weekly questions. I don’t think anyone should try this until they exercise more care . Now I’m stuck without a voice and trying to find help. Thanks Dr Sue 🤬