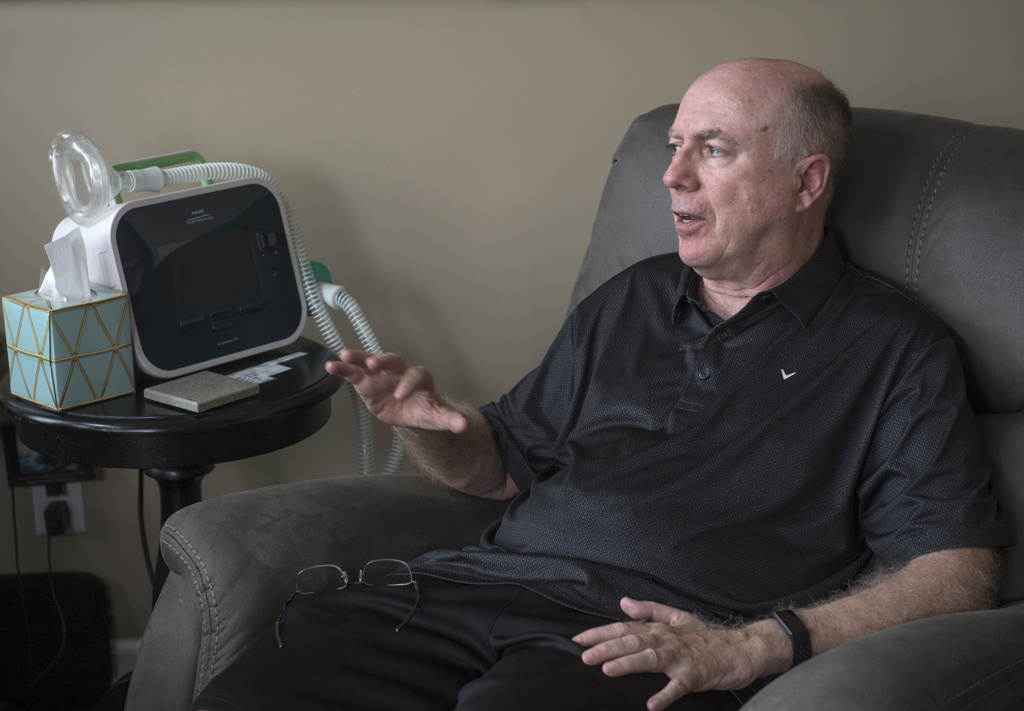
When John Wilkinson sat down with his neurologist, he did not wait to hear his diagnosis.
Before the doctor could speak, John jumped in with the hard truth.
“I have ALS,” he said. “I have two to five years to live. There is no cure.”
Months of mysterious symptoms, visits to several doctors and test results led John and—his neurologist—to the diagnosis.
That was 18 months ago.
There is still no cure for amyotrophic lateral sclerosis—known as ALS or Lou Gehrig’s disease.
But John hopes an innovative new research program will speed the search for effective treatments.
In February, John became the first person at Spectrum Health to take part in the HEALEY ALS Platform Trial, a national study led by the Sean M. Healey & AMG Center for ALS at Massachusetts General Hospital. Spectrum Health is one of 54 sites of the Northeast ALS Consortium approved by the U.S. Food and Drug Administration to administer the trial drug regimens.
A platform trial allows researchers to test multiple treatments simultaneously. This cuts the trial time by half and reduces the cost by 30%.
And people who take part in a platform trial are more likely to receive an investigational drug—instead of a placebo—than in a typical clinical trial.
The platform trial model has been used in cancer research, but this is the first time investigators have used the approach to test ALS treatments.
“Since this is a disease that has urgency to it, there has been a clamoring for a better way to move promising compounds and other therapies through the pipeline more efficiently,” said Paul Twydell, DO, the director of neuromuscular medicine for Spectrum Health.
“It is our hope that the trial will bring us closer to the development of advanced treatments for ALS.”
ALS is a lethal disorder that affects the nerves that control the muscles. It affects about 30,000 people in the U.S. and 500,000 worldwide.
Speeding up research is crucial for those with the condition. As John noted to his doctor, people who have ALS live, on average, two to five years after their diagnosis.
Three ALS treatments have received approval from the FDA.
“Only two of them actually slow the disease down,” Dr. Twydell said.
The third medication treats the emotional outbursts that sometimes occur with ALS.
“That is all we have,” he said. “There have been a lot of medicines looked at over the years. And a lot of failures.”
‘The most active person’
John, 64, grew up in Oklahoma and moved to Michigan in the early 1980s for a job in the oil and gas industry. Now retired, he lives in Grand Rapids with his fiancee, Barbara Harvey.
“He used to be the most active person I know,” Barbara said. “He averaged 25,000 steps a day.”
John shook his head and laughed.
“It was only about 19,000 steps a day,” he said.
There is no doubt John loved to be active. On a typical day, he played pickleball for several hours in the morning and then headed to the YMCA for a workout.
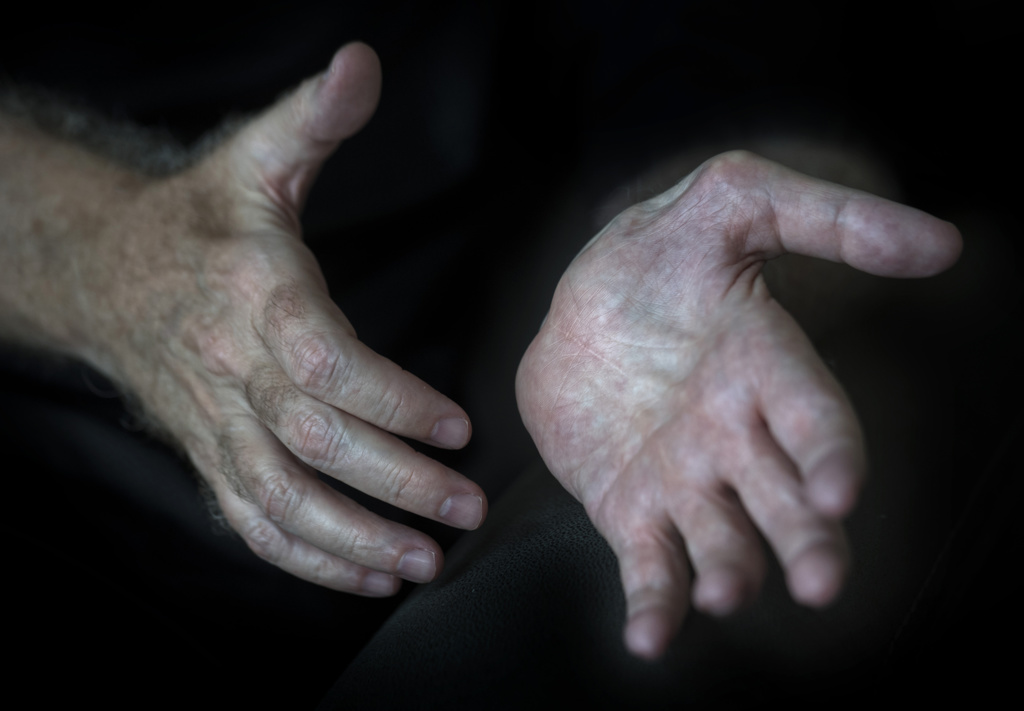
In early 2019, he noticed weakness in his left hand. He underwent carpal tunnel surgery, but his hand strength did not improve. In fact, it grew worse.
After consulting other specialists, he met with a neurologist and underwent an electromyography test to assess his muscles and the motor neurons that control them.
A couple of months later, he had a second EMG to compare with the first.
That test led to the diagnosis of ALS.
He knew little about the disease until that moment. As he researched it, he became overwhelmed by what he learned.
When his neurologist suggested he get a second opinion, John replied: “No. I’ll just pretend like you’re wrong.’”
But John continued to learn all he could about ALS. He receives care from the ALS-Neuromuscular Disease Clinic at Spectrum Health, a multidisciplinary clinic designated a Certified Treatment Center of Excellence by the ALS Association.
The clinic brings together specialists in neurology, respiratory therapy, nutrition, social work, nursing and occupational, speech and physical therapy.
Patients typically meet with the team every three months.
“We address things before they become an issue,” Dr. Twydell said. “We are thinking three to six months ahead of the disease.”
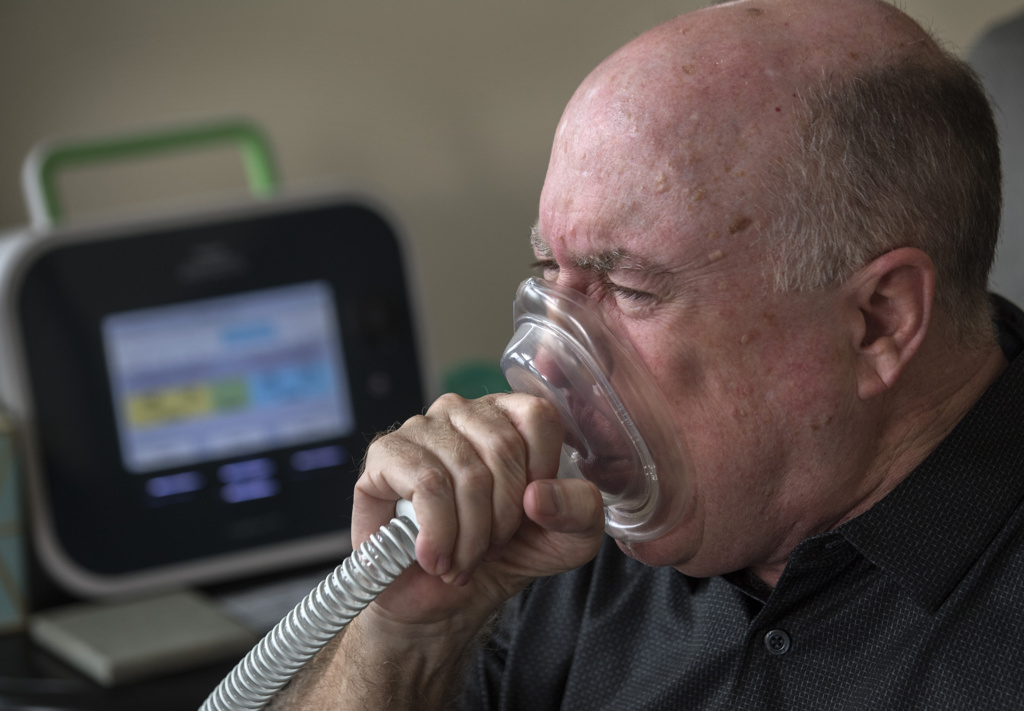
John has equipment to adapt to the changes brought by ALS. Breathing treatments help clear his lungs. He has specially designed plates and silverware that make it easier to eat since he has weakness in both hands now.
He and Barbara recently moved into a condominium with the kitchen, bedroom and living areas all on one floor.
Although he can still walk, a power wheelchair is ready in his den for the day when his mobility is more affected.
He also plans to get a feeding tube, knowing that the disease will continue to make swallowing more difficult.
“We try to live one day at a time and not worry about the future,” Barbara said. “On the other hand, people say to get ahead of this.”
Maintaining that balance is not easy with a disease that causes progressive deterioration of abilities.
“ALS stands for ‘you are always losing something,’” John said.
“There’s never a good time to get a terminal disease diagnosis. It’s hard at first. But you get over it after a while.
“You try to make the best of every day.”
An experimental treatment
Through social media, John connected with other people who have ALS and their caregivers. That is how he learned about the Healey platform trial, which began enrolling its first patients in August 2020.
The research team now is testing four medications, while evaluating other experimental treatments for possible inclusion in the trial.
The medications include: Zilucoplan, a small macrocyclic peptide inhibitor of complement component 5 [C5]; Verdiperstat, an oral myeloperoxidase inhibitor; CNM-Au8 nanocrystalline gold, an intracellular nanocatalyst to support cellular bioenergetics; and pridopidine, a highly selective Sigma-1 receptor.
The trial will include 160 patients in each regimen. Patients are randomly assigned to receive either the investigative treatment or a placebo for 24 weeks.
In a typical clinical trial, half the patients receive a placebo and half receive the investigational medication.
In the Healey platform trial, for every three patients receiving active treatment, one additional patient receives a placebo.
For John, that means, “There is a 75% chance that I have the drug.”
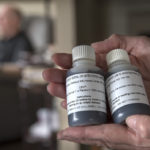
On a table in his kitchen, John has a box that holds 32 smaller boxes. Inside each one are two small bottles containing a dark liquid—either the investigational drug, CNM-Au8 nanocrystalline gold, or a placebo.
Every morning, he pours the liquid into a wineglass and drinks it.
“It tastes like paper,” he said.
At first, John hoped he was taking the investigational drug—not the placebo. After 14 weeks, he is not so sure.
“As it turns out, I am progressing, which in ALS is a bad word,” he said. “I have almost changed my opinion. I kind of hope I have the placebo because the real drug might be better.”
After their 24 weeks in the clinical trial ends, patients have the option to take the experimental drug. And that is another unusual feature of the Healey trial—and a big plus for John.
“We need a cure or better treatments. It was 80 years ago that Lou Gehrig had this,” he said, referring to the New York Yankees baseball player who died of ALS in 1941. “And we are as bad off as he was 80 years ago—in terms of a cure.”
It is good that researchers are evaluating other treatments through the Healey platform trial, he said.
“The Healey trial is so much better than every other trial,” he said. “We are lucky that Spectrum Health is involved. It gives people hope.”















 /a>
/a>
 /a>
/a>
 /a>
/a>
So thankful that John can participate in this trial. Medical advancements such as this one is a ray of hope for our beloved friends, John and Barbara.
Barbara is my mom, and this was such a devastating diagnosis to learn. I’m so glad John had maintained a positive outlook, and my mom has been such a support. I am thankful that science is moving in the right direction and I hope the trial drug shows the signs of improvement John so desperately needs.
This is a very touching story. I first learned of ALS about 20 years ago. I worked for a Neurologist and I remember the first patient we seen with this horrible diagnosis. I seen the progression each time he came in. I pray everyday for a cure for John and others who have ALS. So many years have passed and seems like there should be a good solid treatment by now. John and Barbara you are in my prayers everyday. You both have inspired me to live in the moment and try to put a smile on my face each day. Love you both! Keep fighting John!
My heart soars that my cousin John is in this trial medication!!!!! My prayers for all who have this disease will be rid of it in years to come❤️
This is very encouraging but also so discouraging that so little has been done in 80 years! I’ve known Barbara and Eve for some time, and it is my hopes and prayers that John will see improvements and results in these treatments and make a difference in the lives of others! (A side story: Dogs suffer from a disease called Degenerative Myelosis (sp?) which I’ve been told is the same disease and we lost a dog to this) (not meant to minimize ALS or John and his very difficult and important journey!!)
John is an inspiration to so many!
He has ALWAYS had a positive outlook on life
Stay strong, my friend
John is courageous, and an inspiration!! His sense of humor is a delight to all of us. I am so glad that he has had the opportunity to participate in this trial, for the good of himself and the greater good.
John, you are the most determined man I know. And I’m so happy you have a great women and many friends by your side to help you fight! You are all in my prayers daily.
My friend, Mary, passed away from ALS. She always said it stands for Almighty Living Savior. That was her hope through it all. I hope you do well on the drug John!
I think this is a really good topic. You make this information interesting and engaging. You give readers a lot to think about and I appreciate that kind of post